 
If the 25.00 mL sample that was titrated had been produced by dilution of a 10.If 37.63 mL of thiosulfate solution were needed to titrate a 25.00 mL sample of an acid, what was the H + ion concentration of the acid? The thiosulfate solution was determined to be 1.023 M.The endpoint is reached when the solution becomes colorless. The amount of iodine produced from this reaction can be determined by subsequent titration with thiosulfate: 2S 2O 3 2+ + I 2 → S 4O 6 2− + 2I − Starch is used as the indicator in this titration because starch reacts with iodine in the presence of iodide to form an intense blue complex. ♦ A method for determining the molarity of a strongly acidic solution has been developed based on the fact that a standard solution of potassium iodide and potassium iodate yields iodine when treated with acid: IO 3 − + 5I − + 6H + → 3I 2 + 3H 2O If the overall reaction sequence was 75% efficient, how many grams of carbonic acid were initially dissolved in the 250 mL to produce the calcium bicarbonate?.If 8.5 g of calcium carbonate were obtained by evaporating 250 mL of a solution of calcium bicarbonate followed by drying, what was the molarity of the initial calcium bicarbonate solution, assuming complete reaction?.The reprecipitated calcium carbonate forms a hard scale, or incrustation, on the surface of the object. This weakly acidic carbonic acid solution dissolves marble, converting it to soluble calcium bicarbonate: CaCO 3(s) + H 2CO 3(aq) → Ca(HCO 3) 2(aq)Įvaporation of water causes carbon dioxide to be driven off, resulting in the precipitation of calcium carbonate: Ca(HCO 3) 2(aq) → CaCO 3(s) + H 2O(l) + CO 2(g) Groundwater dissolves atmospheric carbon dioxide to produce an aqueous solution of carbonic acid: CO 2(g) + H 2O(l) → H 2CO 3(aq) Under normal circumstances, carbonic acid decomposes into CO 2 and H 2 O. However, carbonic acid can only exist at very low concentrations. Do tests for carbon dioxide gas.♦ Reactions that affect buried marble artifacts present a problem for archaeological chemists. Step 3: The reaction is the combination of bicarbonate ions and hydrogen ions that will first form carbonic acid (H 2 CO 3). Add dilute acid to the carbonate compound.There are so many tests to identify carbonate ion. What test is done to identify carbonate ions Compare these pH values with pH of strong bases. Metal carbonate solutions are weak bases and have pH values just higher than 7 (like 8,9,10). PH of dilute solution of carbonate solution Therefore, add aqueous calcium chloride solution to the carbonate ion solution. But this test is not enough to prove the presence of carbonate ion. Metal carbonate compounds are usually weak bases. 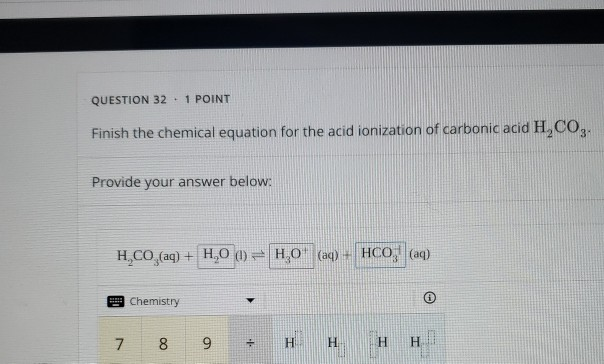 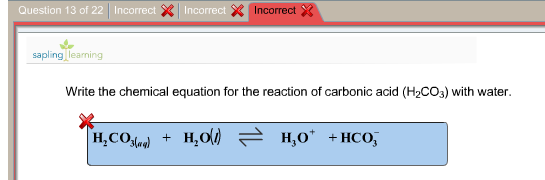
Test the presence of Carbonate ions in waterĬheck pH value. When excess gas is sent, white precipitate dissolves.Ĭolours of phenolphthalein with CO 3 2- and HCO 3. Take a piece of white solid and add dilute HCl to it. Now you have do an experiment which will varyfy there is carbonate ion. Now another student says anion should be carbonate ion. But the acidic oxide is not completely neutralized in this case. Thus, only when the concentration of the alkali solution is quite low, the reaction proceeds via the formation of carbonic acid. You are provided a white solid and one student has found there is calcium metal as the cation. ABSTRACT: Wellbore integrity of abandoned wells is of high priority for ensuring the containment of sequestered CO2. N a O H + C O X 2 N a H C O X 3 N a H C O X 3 + N a O H N a X 2 C O X 3 + H X 2 O. Testing for carbonate ions in calcium carbonate Reactions of bicarbonate with organic compoundsĬarboxylic acid reacts with bicarbonate ion to give carbon dioxide. So you can identify the two compounds magnesium carbonate and barium carbonate when forming final white precipitate is given. Then add little amount of sulfuric acid ( H 2SO 4 ). White precipitates dissolve and give colourless solutions. We know all alkali earth metal carbonate compounds gives white precipitates in water.įirst we add dilute HCl to MgCO 3 and CaCO 3. How to identify magnesium carbonate and barium carbonate But, BaSO 4 is insoluble in dilute HCl and exists as the white precipitate. So we have to do some reactions which will affect differently, for carbonate and sulfate ion. How to identify BaCO 3 and BaSO 4 precipitates?īoth BaCO 3 and BaSO 4 are white precipitates Ba 2+ ion is common and anion is different for both compounds. You can see white precipitate dissolve in water and a gas is emmitted from the solution. Then add dilute HCl acid solution to the precipitate. Separate the precipitate from the solution. Barium carbonate (BaCO 3), white precipitate is given. Potassium will give purple colour to flame test.Īdd barium chloride (BaCl 2) solution to carbonate ion solution. To test potassium ion, you can do flame test. Potassium carbonate is soluble in water and a colourles solution. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
